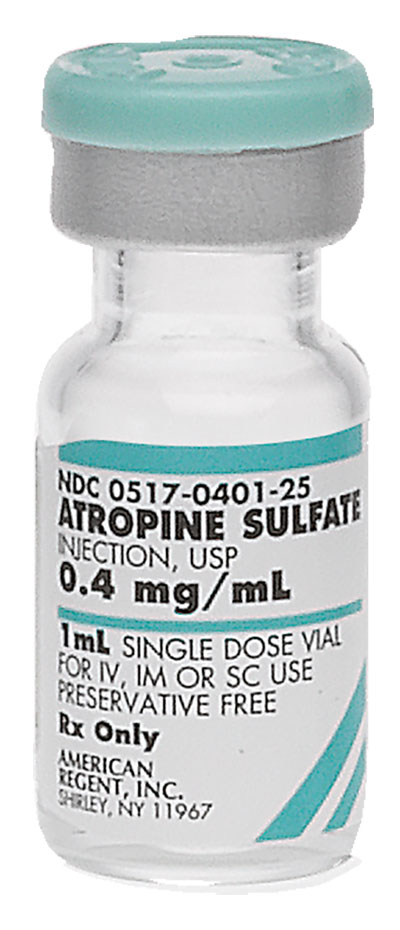
Pack NDC#
0517-0401-25
|
Atropine Sulfate Injection, USP (Marketed Unapproved) †
|
0.4 mg/mL
|
1 mL
Single Dose Vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
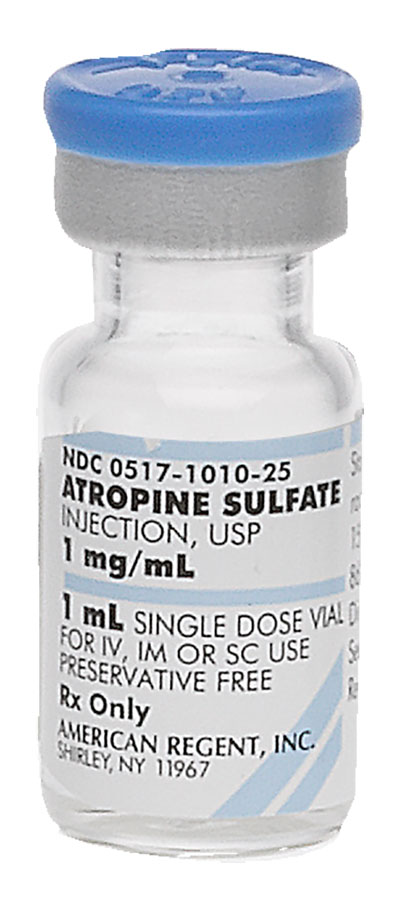
Pack NDC#
0517-1010-25
|
Atropine Sulfate Injection, USP (Marketed Unapproved) †
|
1 mg/mL
|
1 mL
Single Dose Vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
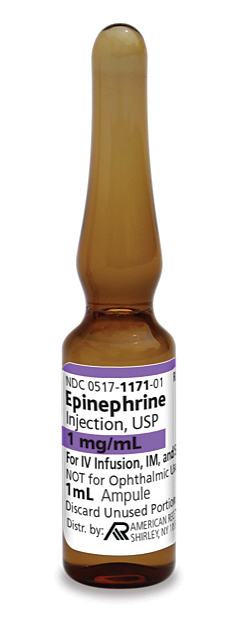
Pack NDC#
0517-1171-10
|
Epinephrine Injection, USP
|
1 mg/mL
|
1 mL single-dose ampule
|
10 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
0517-2901-01

|
Gvoke VialDxTM (glucagon injection)
|
0.45 mg/mL
|
1 mg per 0.2 mL
Single-dose vial
|
1 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
0517-2901-10

|
Gvoke VialDxTM (glucagon injection)
|
0.45 mg/mL
|
1 mg per 0.2 mL
Single-dose vial
|
10 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
0517-3005-25

|
Sterile Water For Injection, USP †
|
—
|
5 mL
Single Dose Vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
0517-3010-25

|
Sterile Water For Injection, USP †
|
—
|
10 mL
Single-dose Vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
0517-3020-25

|
Sterile Water For Injection, USP †
|
—
|
20 mL
Single-dose Vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
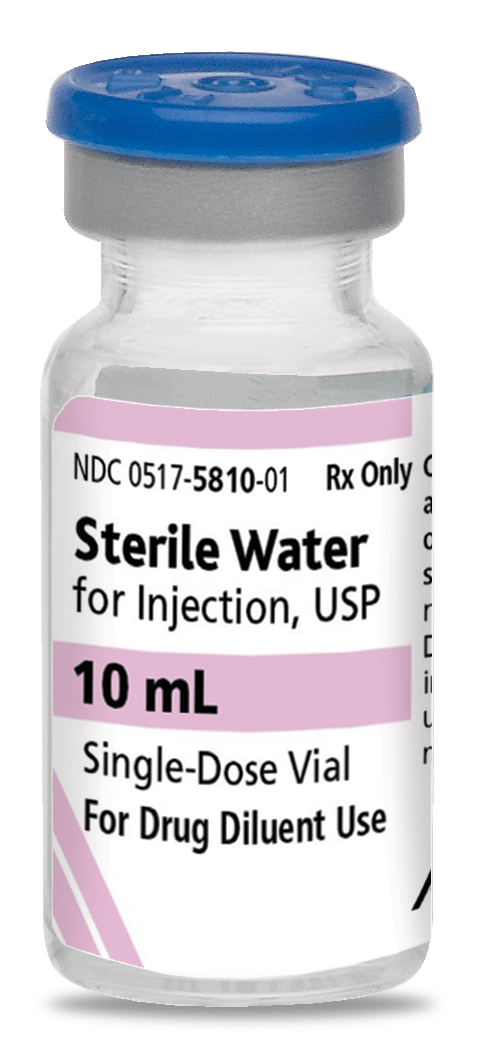
Pack NDC#
0517-5810-25
|
Sterile Water For Injection, USP
|
-
|
10 mL
Glass fliptop single-dose vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
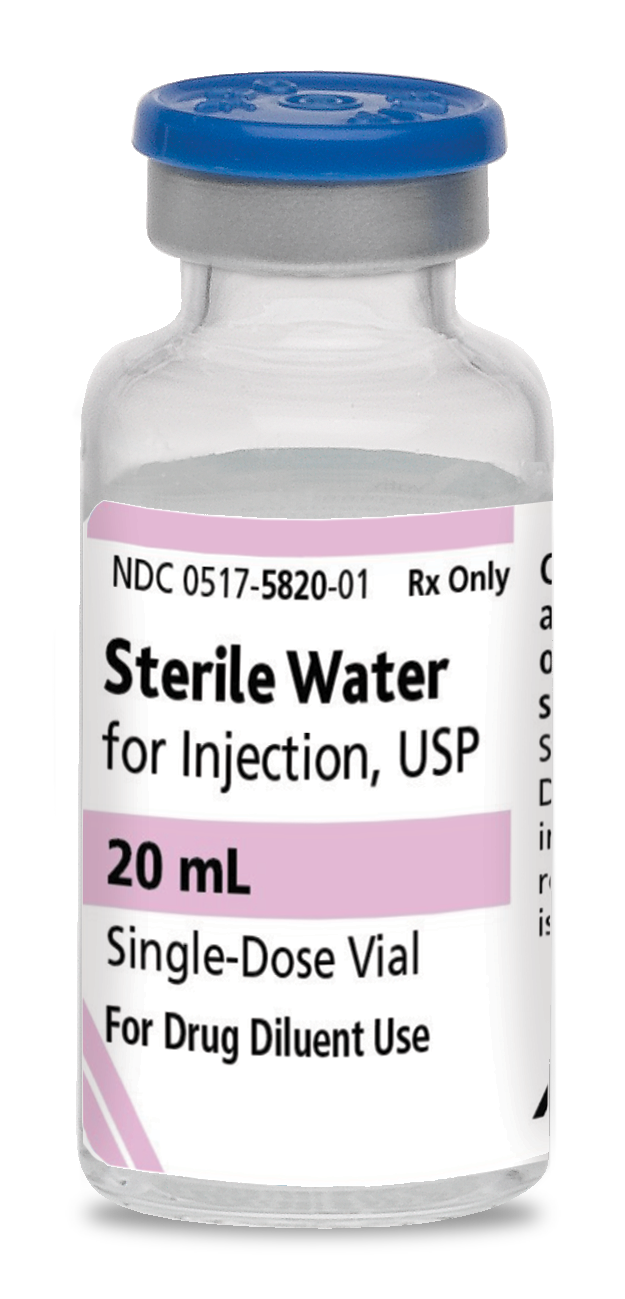
0517-5820-25
|
Sterile Water For Injection, USP
|
-
|
20 mL
Glass fliptop single-dose vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|
Pack NDC#
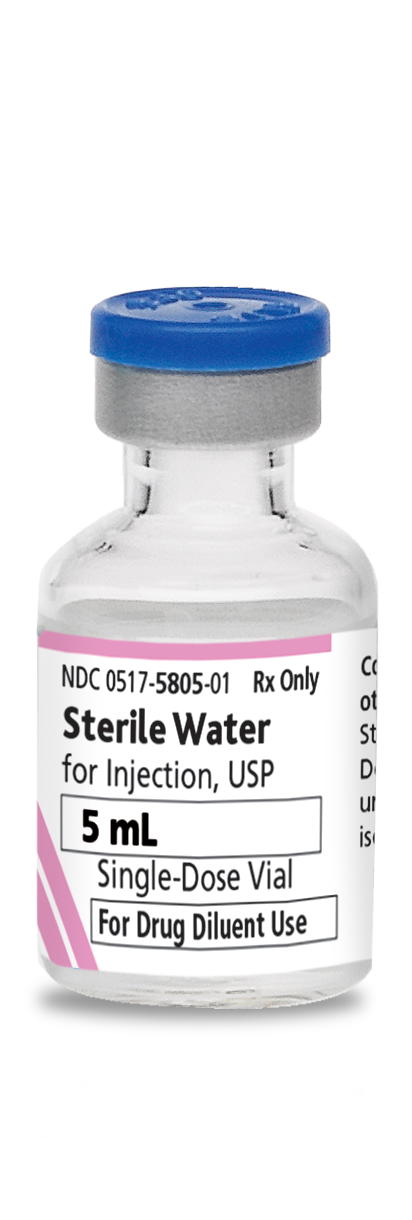
0517-5805-25
|
Sterile Water For Injection, USP
|
-
|
5 mL
Glass fliptop single-dose vial
|
25 |
Product Label
Full Prescribing Information
Safety Data Sheet
|
Discontinued
|









