Contact Us
By clicking Submit, you confirm that you accept our Privacy Policy and that you agree to your personal contact information being used to contact you and added to our database. If at any time you wish your personal information to be removed from the American Regent® database, please submit a message request to corpcommunications@americanregent.com.
|
WARNING: SEROTONIN SYNDROME WITH CONCOMITANT USE OF SEROTONERGIC DRUGS AND OPIOIDS ProvayBlue® may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs and opioids. Avoid concomitant use of ProvayBlue® with selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), and monoamine oxidase inhibitors (MAOIs) and opioids. |
CONTRAINDICATIONS
ProvayBlue® is contraindicated in patients with severe hypersensitivity reactions to methylene blue or any other thiazine dye, and in patients with glucose-6-phosphate dehydrogenase deficiency (G6PD) due to the risk of hemolytic anemia.
WARNINGS AND PRECAUTIONS
Serotonin Syndrome with Concomitant Use of Serotonergic Drugs and Opioids
The development of serotonin syndrome has been reported with the use of methylene blue class products. Most reports have been associated with concomitant use of serotonergic drugs (eg, selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), monoamine oxidase inhibitors). Opioids and dextromethorphan may increase the risk of developing serotonin syndrome. Some of the reported cases were fatal. Symptoms associated with serotonin syndrome may include the following combination of signs and symptoms: mental status changes (eg, agitation, hallucinations, delirium, and coma), autonomic instability (eg, tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, and hyperthermia), neuromuscular symptoms (eg, tremor, rigidity, myoclonus, hyperreflexia, and incoordination), seizures, and/or gastrointestinal symptoms (eg, nausea, vomiting, diarrhea). Avoid concomitant use of ProvayBlue® with serotonergic drugs and opioids.
Patients treated with ProvayBlue® should be monitored for the emergence of serotonin syndrome. If symptoms of serotonin syndrome occur, discontinue use of ProvayBlue®, and initiate supportive treatment. Inform patients of the increased risk of serotonin syndrome and advise them not to take serotonergic drugs within 72 hours after the last dose of ProvayBlue®.
Hypersensitivity
Anaphylactic reactions to methylene blue class products have been reported. If anaphylaxis or other severe hypersensitivity reactions (eg, angioedema, urticaria, bronchospasm) should occur, discontinue use of ProvayBlue® and initiate supportive treatment. ProvayBlue® is contraindicated in patients who have experienced anaphylaxis or other severe hypersensitivity reactions to a methylene blue class product in the past.
Lack of Effectiveness
Methemoglobinemia may not resolve or may rebound after response to treatment with ProvayBlue® in patients with methemoglobinemia due to aryl amines such as aniline or sulfa drugs such as dapsone. Monitor response to therapy with ProvayBlue® through resolution of methemoglobinemia. If methemoglobinemia does not respond to 2 doses of ProvayBlue® or if methemoglobinemia rebounds after a response, consider additional treatment options.
Patients with G6PD deficiency may not reduce ProvayBlue® to its active form. ProvayBlue® may not be effective in patients with G6PD deficiency.
Hemolytic Anemia
Hemolysis can occur during treatment of methemoglobinemia with ProvayBlue®. Laboratory testing may show Heinz bodies, elevated indirect bilirubin, and low haptoglobin, but the Coombs test is negative. The onset of anemia may be delayed 1 or more days after treatment with ProvayBlue®. The anemia may require red blood cell transfusions. Use the lowest effective number of doses of ProvayBlue® to treat methemoglobinemia. Discontinue ProvayBlue® and consider alternative treatments of methemoglobinemia if severe hemolysis occurs.
Treatment of patients with G6PD deficiency with ProvayBlue® may result in severe hemolysis and severe anemia. ProvayBlue® is contraindicated for use in patients with G6PD deficiency.
Interference with In Vivo Monitoring Devices
The presence of methylene blue in the blood may result in an underestimation of the oxygen saturation reading by pulse oximetry.
A fall in the Bispectral Index (BIS) has been reported following administration of methylene blue class products. If ProvayBlue® is administered during surgery, alternative methods for assessing the depth of anesthesia should be employed.
Effects on Ability to Drive and Operate Machinery
Treatment with ProvayBlue® may cause confusion, dizziness, and disturbances in vision. Advise patients to refrain from driving or engaging in hazardous occupations or activities such as operating heavy or potentially dangerous machinery until such adverse reactions to ProvayBlue® have resolved.
Interference with Laboratory Tests
ProvayBlue® is a blue dye which passes freely into the urine and may interfere with the interpretation of any urine test which relies on a blue indicator, such as the dipstick test for leucocyte esterase.
ADVERSE REACTIONS
The safety of ProvayBlue® in adults with acquired methemoglobinemia was assessed in 31 patients who received at least 1 dose of ProvayBlue®. Most doses administered were 1 mg/kg (82.9%), but doses from 0.78 mg/kg to 2 mg/kg were administered. All patients received at least one dose of ProvayBlue®; two received two doses, and one received three doses. Serious adverse reactions occurred in 3.2% of patients who received ProvayBlue®.
A serious adverse reaction of seizure-like phenomenon was reported in one patient. Adverse reactions (≥2%) included headache, hypokalemia, diarrhea, hypomagnesemia, myoclonus, nausea, and seizure-like phenomena.
The safety of ProvayBlue® in pediatric patients with acquired methemoglobinemia was assessed in two retrospective case series that included two pediatric patients treated with ProvayBlue® and 12 treated with another methylene blue product. The case series included patients in the following age groups: 3 neonates (<1 month), 4 infants (1 month to <2 years), 4 children (2 years to <12 years), and 3 adolescents (12 years to <17 years). The safety profile in pediatric patients was similar to that in adult patients.
Other adverse reactions reported to occur following administration of methylene blue class products include, but are not limited to, the following: hemolytic anemia, hemolysis, hyperbilirubinemia; palpitations, tachycardia; eye pruritus, ocular hyperemia, vision blurred; abdominal pain lower, dry mouth, flatulence, glossodynia, tongue eruption; death; infusion site extravasation, infusion site induration, infusion site pruritus, infusion site swelling, infusion site urticaria, peripheral swelling; thirst; elevated liver enzymes; myalgia; dysuria; nasal congestion, oropharyngeal pain, rhinorrhea, sneezing; necrotic ulcer, papule, phototoxicity; and hypertension.
DRUG INTERACTIONS
The concomitant use of ProvayBlue® with other drugs that affect the serotonergic neurotransmitter system has resulted in serotonin syndrome. Although the mechanism is not clearly understood, literature reports suggest ProvayBlue® is a potent reversible inhibitor of monoamine oxidase. Avoid concomitant use of ProvayBlue® with medicinal products that enhance serotonergic transmission including antidepressants like SSRIs (selective serotonin reuptake inhibitors), SNRIs (serotonin and norepinephrine reuptake inhibitors), MAOIs (monoamine oxidase inhibitors), bupropion, buspirone, clomipramine, mirtazapine, linezolid, opioids, and dextromethorphan because of the potential for serious CNS reactions, including potentially fatal serotonin syndrome. If the intravenous use of ProvayBlue® cannot be avoided in patients treated with serotonergic medicinal products, choose the lowest possible dose and observe the patient closely for CNS effects for up to 4 hours after administration.
USE IN SPECIFIC POPULATIONS
Pregnancy and Lactation
ProvayBlue® may cause fetal harm when administered to a pregnant woman. Intra-amniotic injection of pregnant women with a methylene blue class product during the second trimester was associated with neonatal intestinal atresia and fetal death. Advise pregnant women of the potential risk to the fetus.
There is no information regarding the presence of methylene blue in human milk. Because of the potential for serious adverse reactions, including genotoxicity, discontinue breast-feeding during, and for up to 8 days after, treatment with ProvayBlue®.
Geriatric Use
Because elderly patients are more likely to have decreased renal function, treatment of methemoglobinemia in these patients should use the lowest number of doses needed to achieve a response.
Renal Impairment
Methylene blue concentrations increased in subjects with renal impairment (eGFR 15 to 89 mL/min/1.73 m2). Adjust ProvayBlue® dosage in patients with moderate or severe renal impairment (eGFR 15 to 59 mL/min/1.73 m2). No dose adjustment is recommended in patients with mild renal impairment (eGFR 60 – 89 mL/min/1.73 m2).
Hepatic Impairment
Methylene blue is extensively metabolized in the liver. Monitor patients with any hepatic impairment for toxicities and potential drug interactions for an extended period of time following treatment with ProvayBlue®.
OVERDOSAGE
In case of overdose of ProvayBlue®, maintain the patient under observation until signs and symptoms have resolved, monitor for cardiopulmonary, hematologic and neurologic toxicities, and institute supportive measures.
DOSAGE AND ADMINISTRATION
Preparation and Storage
ProvayBlue® is hypotonic and may be diluted before use in a solution of 50 mL 5% Dextrose Injection in order to avoid local pain, particularly in the pediatric population. Use the diluted solution immediately after preparation.
Avoid diluting with sodium chloride solutions, because it has been demonstrated that chloride reduces the solubility of methylene blue.
INDICATIONS AND USAGE
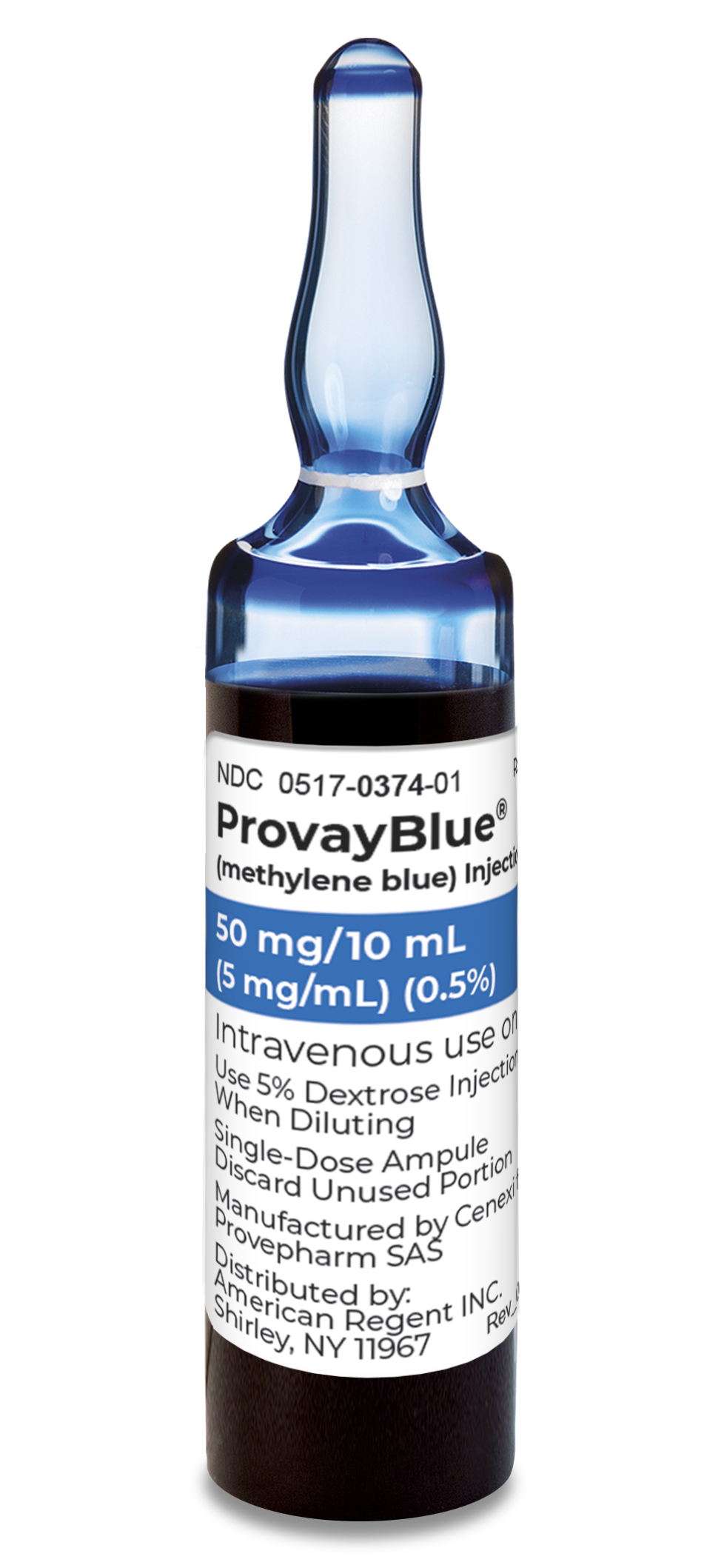
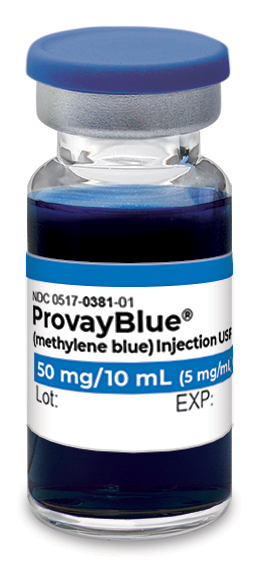
ProvayBlue® (methylene blue) is indicated for the treatment of pediatric and adult patients with acquired methemoglobinemia.
For additional safety information, including BOXED WARNING, please see Full Prescribing Information.
You are encouraged to report Adverse Drug Events to American Regent, Inc. at 1-800-734-9236 or to the FDA by visiting www.fda.gov/medwatch or calling 1-800-FDA-1088.
REF-0131 2/2024