Contact Us
By clicking Submit, you confirm that you accept our Privacy Policy and that you agree to your personal contact information being used to contact you and added to our database. If at any time you wish your personal information to be removed from the American Regent® database, please submit a message request to corpcommunications@americanregent.com.
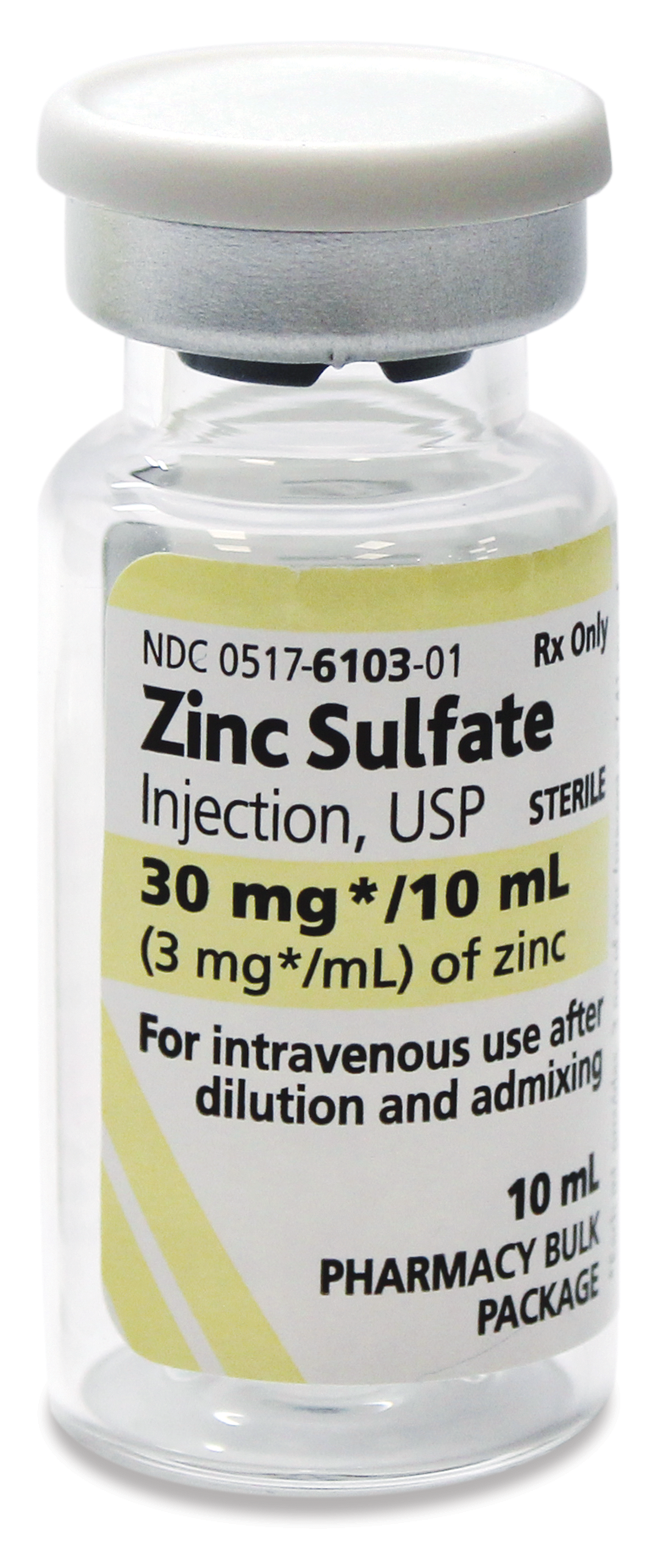
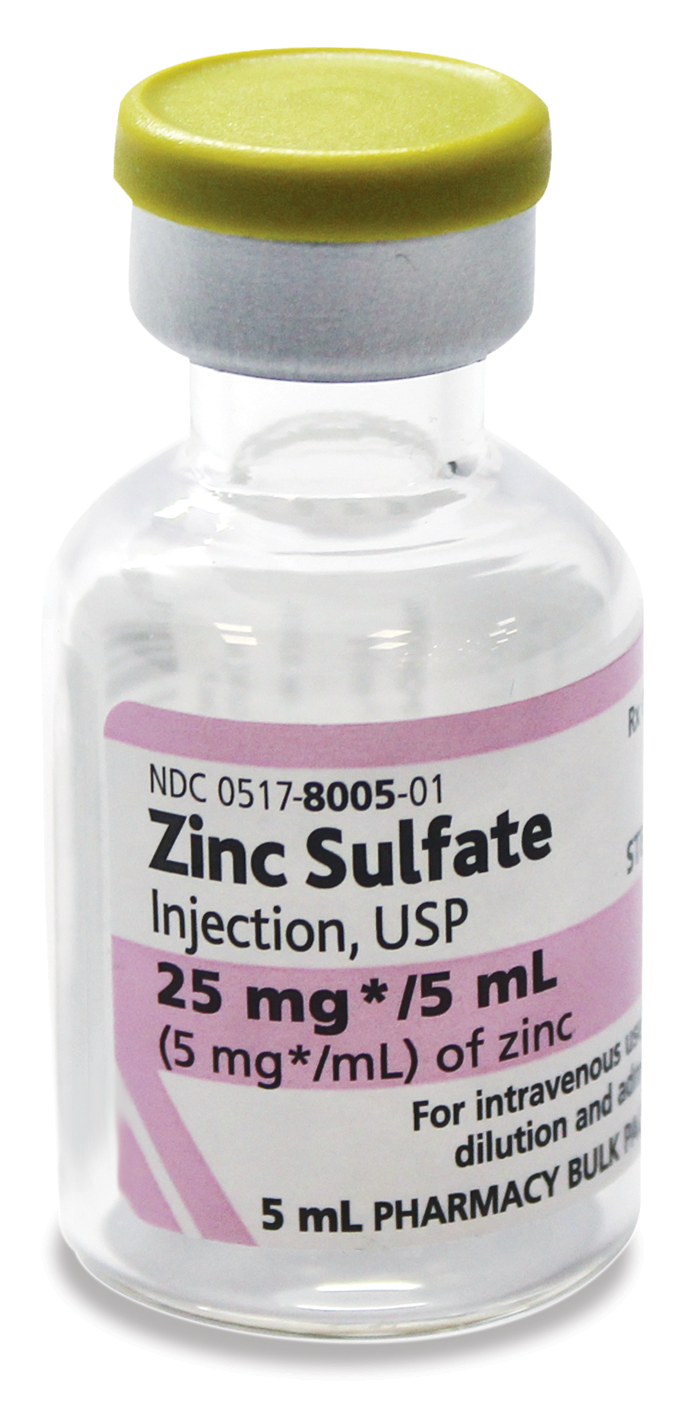
For intravenous use
INDICATIONS AND USAGE
Zinc Sulfate is a trace element indicated in adult and pediatric patients as a source of zinc for parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated.
IMPORTANT ADMINISTRATION INFORMATION
Zinc Sulfate Injection is supplied as a pharmacy bulk package for admixing use only. It is not for direct intravenous infusion.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Zinc Sulfate Injection is contraindicated in patients with known hypersensitivity to zinc.
WARNINGS AND PRECAUTIONS
Pulmonary Embolism due to Pulmonary Vascular Precipitates: If signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. The infusion set and catheter should be checked periodically for precipitates.
Vein Damage and Thrombosis: Zinc Sulfate Injection has a low pH and must be prepared and used as an admixture in PN solutions. Solutions with osmolarity of 900 mOsm/L or more must be infused through a central venous catheter.
Aluminum Toxicity: Zinc Sulfate Injection contains aluminum that may be toxic. Aluminum may reach toxic levels with prolonged parenteral administration if kidney function is impaired. Preterm infants are particularly at risk for aluminum toxicity because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which also contain aluminum.
Monitoring and Laboratory Tests: Monitor Zinc concentrations, fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count and coagulation parameters throughout treatment.
Copper Deficiency: Several post-marketing cases have reported that high doses of supplemental zinc (approximately 10 times the recommended dosage of 3 mg/day Zinc Sulfate Injection in adults) taken over extended periods of time (i.e., months to years) may result in decreased enteral copper absorption and copper deficiency.
Hypersensitivity Reactions: If hypersensitivity reactions occur, discontinue Zinc Sulfate Injection and initiate appropriate medical treatment.
ADVERSE REACTIONS
No zinc-related adverse reactions have been reported in clinical studies or postmarketing reports in patients receiving intravenously administered PN solutions containing zinc sulfate within the recommended dosage range.
USE IN SPECIFIC POPULATIONS
Pregnancy: Risk Summary: Administration of the recommended dose of Zinc Sulfate Injection in PN is not expected to cause major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Lactation: Risk Summary: Zinc is present in human milk. There is no information on the effects of zinc sulfate on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Zinc Sulfate Injection and any potential adverse effects on the breastfed infant from Zinc Sulfate Injection or from the underlying maternal condition.
Pediatric Use: Safety and dosing recommendations in pediatric patients are based on published literature describing controlled studies of zinc-containing products in pediatric patients.
Geriatric Use: Dose selection should be individualized based on the patient’s clinical condition, nutritional requirements, and additional nutritional intake provided orally or enterally to the patient.
OVERDOSAGE
There are reported cases of overdosage with intravenous zinc in parenteral nutrition.
For additional safety information, please see Full Prescribing Information.
You are encouraged to report Adverse Drug Events to American Regent, Inc. at 1-800-734-9236 or to the FDA by visiting www.fda.gov/medwatch or calling 1-800-FDA-1088.
REF-1299 10/2020